It comprises one sigma, one delta, and two pi bonds.It is mainly seen in the axis that is present between atoms.Bonds of four are rare to form and see.The structure of three bonds contains two sigma bonds and one perpendicular axis atom on the orbitals.The Eugene bond structure holds one pi and one sigma bond.The constant structure of the bond contains one pi bond and one sigma bond.Pi bond, sigma bond, and delta bond are the three different structures of multiple bonds. What is the Pi Bond structure in multiple bonds? So, as a result, the pi bond holds the lower degree of overlapping orbitals in its atom structure, resulting in a weaker bonding structure. Pi bond strength is measured in a quantum mechanical way and compared to the sigma bond strength. Considering the single c-c bond holds more strength than the double c-c bond that accounts for one pi and one sigma bond.Īccording to bond strength, it does not add fragility as a sigma bond as pi bonds have weak stability. The Sigma bond is not that fragile like pi bonds and forms a particular strength to pi bonds. This tells that the atom in the pi link does not rotate around the other atoms. This is only reasonable if all of the atoms in the molecule are in the exact plane. The pi becomes strong if the p orbitals have complete sideways overlapping and are parallel to each other. The pi gets weak due to the partially lateral orbit formation. If electrons heavily influence the axis bond on the p orbitals, it forms a pi bond. What are Pi Bond Formations?Īs per the pi bond definition, two laterally or bonding sideways bonds are called lateral p orbitals, and the formation is called the pi orbital or covalent pi bond. D orbitals can also be placed in this bonding. The pi bond definition says that pi is related to the p orbitals as it has a parallel arrangement of atoms. The pi bond definition is a naturally covalent chemical bond that involves the overlapping lateral structure of orbitals belonging to different atoms. Unsaturated molecules hold the pi bond mostly.It is less reactive to bond formations. 
The pi bond decides the length of the molecule formations.It is not the prime bond in the information that is formed later. 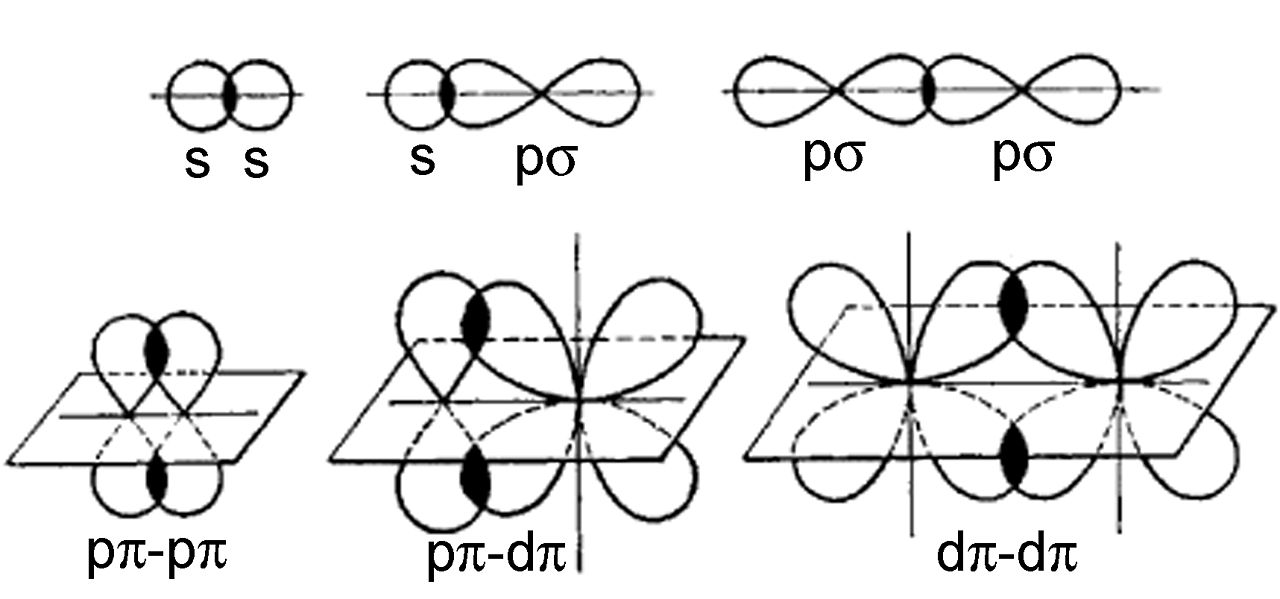
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
